What if cognitive aging isn't about losing brain cells,
but losing the right timing?
Decoherence via
Demyelination
A Hypothesized Mechanism of Cognitive Decline
ages 40 – 99
tracts analyzed
regions mapped
This paper is currently under review. Please enter the password to continue.
What if cognitive aging isn't about losing brain cells,
but losing the right timing?
A Hypothesized Mechanism of Cognitive Decline
Your brain is not a single computer. It is a network of roughly 86 billion neurons organized into dozens of specialized regions, each handling a different job, from recognizing faces to planning tomorrow. Thinking, remembering, and deciding require many of these regions to activate at the same time, in precise coordination.
The Concert Hall: Imagine an orchestra whose musicians sit in different buildings across a city, connected only by cables carrying their sound. For the symphony to work, every note must arrive at the mixing board within milliseconds of the right beat. If even one cable introduces a delay, that section falls out of rhythm and the whole piece suffers.
In the brain, those cables are white matter tractsWhite Matter TractsBundles of nerve fibers that connect distant brain regions, like internal cables.
Full glossary entry →: bundles of long nerve fibers that link distant regions. Each fiber is wrapped in a fatty coating called myelinMyelinA fatty insulating coating on nerve fibers that speeds up and stabilizes signals.
Full glossary entry →. Myelin does two things: it speeds up electrical signals (from ~1 m/s to ~100 m/s) and, critically, it keeps their timing precise.
Colored bands = myelin insulation. Gaps in the lower track show where myelin has degraded. The signal pulse travels noticeably slower.
As we age, this myelin coating breaks down, but not uniformly. Tracts serving memory, complex reasoning, and language — for example, the uncinate fasciculus and fornix — degrade fastest. Tracts handling basic vision and movement are largely spared. This uneven pattern explains why an 80-year-old may struggle to find a word mid-sentence yet walk across a room without difficulty.
Two effects combine to produce this pattern: late-developing pathways start with thinner myelin than early-developing ones, and they lose myelin faster with age. Our diffusion-MRI data measure the second — heterogeneous loss rates, not just starting differences. The pattern itself is well-documented in the white-matter aging literature; the DDH takes the heterogeneity as an empirical given and traces its functional consequences for cognition.
Uneven Wear: Imagine the cable to the oboe section fraying while the violin cable stays intact. The oboist still plays perfectly, but their notes arrive a beat late. The audience hears dissonance, not because anyone forgot the music, but because the delivery infrastructure failed selectively.
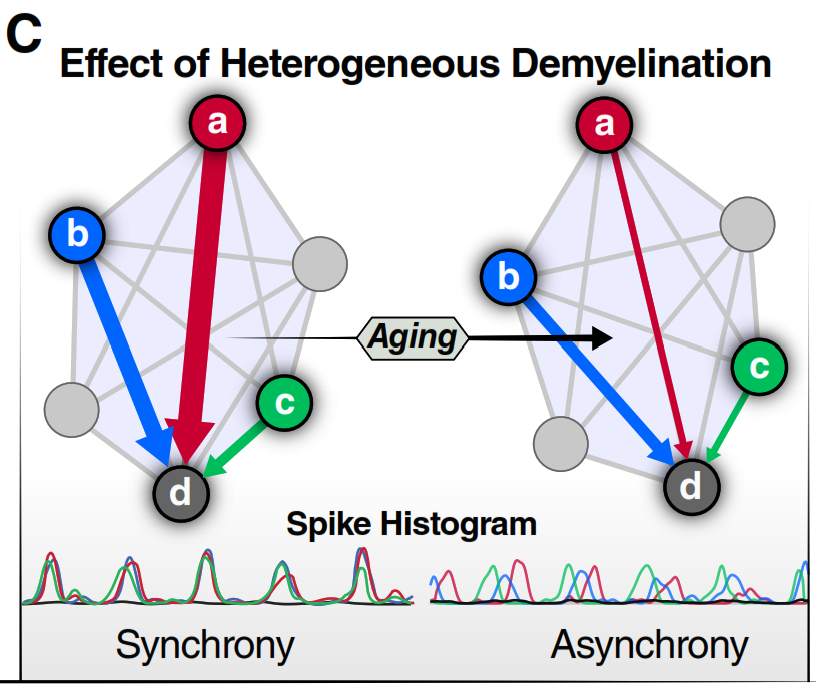
The word coherence means waves moving in lockstep. DecoherenceDecoherenceLoss of synchronization between brain signals, disrupting coordinated network activity.
Full glossary entry → means they have drifted apart. The DDH borrows this concept from physics to describe what happens in the aging brain: different regions need to oscillate in synchronized rhythms (like musicians keeping a shared tempo) to produce thought. When myelin degrades at different rates across different pathways, signals arrive at the wrong phase of these rhythms, and the synchronization breaks down.
The borrowing is metaphorical. DDH describes a classical mechanism — heterogeneous conduction delays in physical wiring — not a quantum-mechanical phenomenon. What both senses share is the loss of phase-coherence among elements of a system.
Higher-order thinking tracts (red/orange) lose myelin much faster than basic sensory & motor tracts (green). This matches the real-world pattern: complex reasoning slows before walking or seeing does.
Schematic of FA (fiber integrity) vs. age for higher-order cognitive tracts. Decline is gradual until ~60, then accelerates sharply. Each line represents a different tract; red tracts are most affected.
Myelin reaches peak thickness. Brain networks synchronize with minimal effort. Cognitive processing speed and mental flexibility are at their lifetime maximum.
Gradual, mostly linear decline begins in select tracts. You may notice slightly slower word retrieval or reduced ease in switching between tasks.
Decline accelerates nonlinearly in many key pathways (a 'hockey-stick' curve). Processing speed drops measurably; multitasking and novel problem-solving become harder.
If the core problem is degrading insulation rather than dying neurons, the therapeutic target changes entirely. The brain's wiring plan remains intact. What fails is the myelin that keeps signals on schedule. This opens a different class of interventions:
The wiring diagram of the brain is still there. We just need to keep its insulation healthy.
Early neuroscience, shaped by studies of focal brain lesions, assumed each function lived in one area. Modern imaging (fMRI, MEG) and large-scale neural recordings have overturned this view. Cognitive operations (attention, memory, reasoning) emerge from coordinated activity across distributed functional networks, not from any single region:
These networks reconfigure dynamically depending on the task. Their proper function depends on inter-regional oscillatory synchronyOscillatory SynchronyBrain regions vibrating in coordinated rhythms to enable communication.
Full glossary entry →: neurons in distant areas must fire in coordinated, phase-locked rhythms (in the gamma, beta, and theta frequency bands). Disrupting this synchrony disrupts the computation itself.
Convergent insight, independent foundations. The importance of timing for cognition has been arrived at from multiple directions. Pascal Fries' Communication Through Coherence framework (Fries 2005, 2015), developed from electrophysiology in primate visual cortex, demonstrates that neuronal groups must synchronize in specific phase relationships for effective signal transfer at the local circuit level. The DDH arrives at a parallel insight from a different starting point entirely: cognitive aging biology, white-matter microstructure, and the population-scale trajectory of myelin loss across the human lifespan. The two theories are mutually compatible and reinforce one another, but neither is derived from the other. Where CTC characterizes how coherence functions in healthy microcircuits, DDH identifies the structural mechanism by which whole-brain timing fails over decades of aging, and offers human MRI evidence that the visual-cortex framework alone cannot provide.
Myelin sheaths enable saltatory conductionSaltatory ConductionElectrical signals jumping between gaps in myelin, traveling up to 100x faster.
Full glossary entry →: electrical signals jump between regularly spaced gaps in the myelin (called Nodes of Ranvier) along the axon, dramatically increasing speed. But speed alone is not enough. Myelin also reduces temporal jitterTemporal JitterRandom variation in signal arrival times; myelin minimizes this variation.
Full glossary entry →: it narrows the variance in when each action potential arrives at its target. This precision is what allows incoming signals to land within the correct excitatory phase window of the target circuit's oscillation.
Even millisecond-scale timing deviations can push incoming signals from an excitatory phase window into an inhibitory one, fundamentally disrupting inter-regional communication. Myelin precision is not a luxury; it's a computational necessity.
The target circuit oscillates (top). A myelinated signal (middle) arrives in phase with the excitatory window (green zone). A demyelinated signal (bottom) arrives late, hitting the inhibitory window (red zone), and the message is effectively blocked.
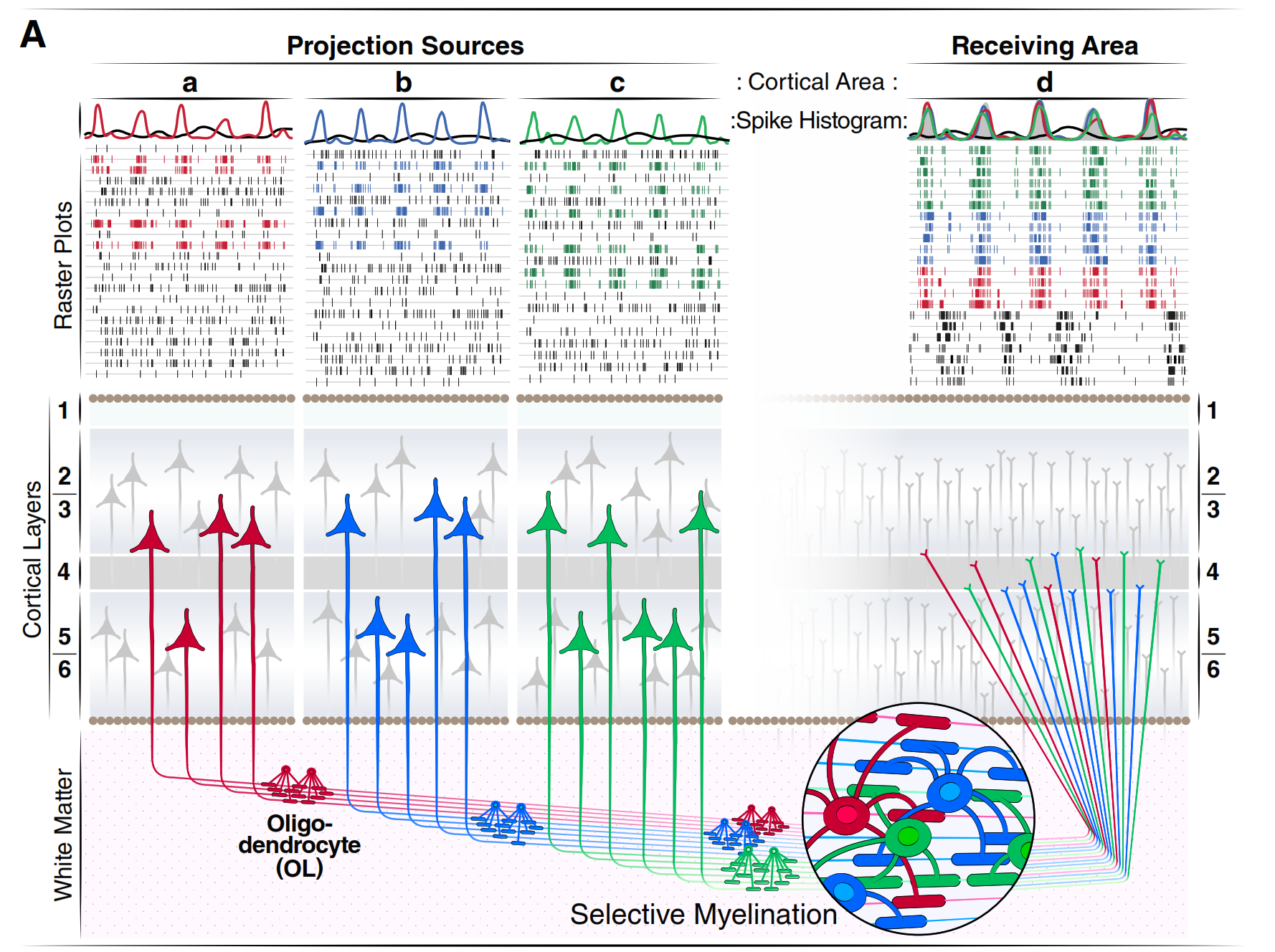
Critically, myelination is not static. Myelin plasticity — the brain's ability to actively add, thin, or remodel myelin in response to use — provides a second learning system alongside Hebbian plasticityHebbian Plasticity'Neurons that fire together wire together': learning at the synapse level.
Full glossary entry →:
Operates at the junctions between neurons (synapses), strengthening or weakening individual connections based on activity.
Operates along the cables between regions (axons), tuning how fast and how precisely signals travel. Oligodendrocytes build "smart wiring": active axons become more heavily myelinated.
Evidence for myelin plasticity is direct: blocking oligodendrocyte differentiation prevents mice from learning new motor skills. Conversely, motor learning triggers oligodendrocyte proliferation. In humans, both working memory and episodic memory require the generation of new myelinating oligodendrocytes, establishing myelin plasticity as essential for cognition, not just motor function.
If the DDH is correct, the human aging brain should show three specific structural signatures. The paper tests each one, in order, using diffusion-weighted MRI from 638 participants (ages 40–99) in the UCSF Hillblom Aging Network cohort.
What this study shows — and what it doesn't. A cross-sectional cohort of 638 humans, each scanned and cognitively tested once. The three findings below — heterogeneous tract decline, microstructure consistent with myelin loss, and a single dominant brain–cognition axis — are internally consistent with the DDH and replicate patterns reported in the white-matter aging literature. They are not, by themselves, proof of it. Definitive causal mechanism would require longitudinal data within individuals, interventional remyelination studies, or Mendelian randomization isolating myelin-relevant genetic variation. None of those exist yet. What the structural data establish is that any complete theory of normal cognitive aging must account for this pattern — and the DDH is the simplest theory that does.
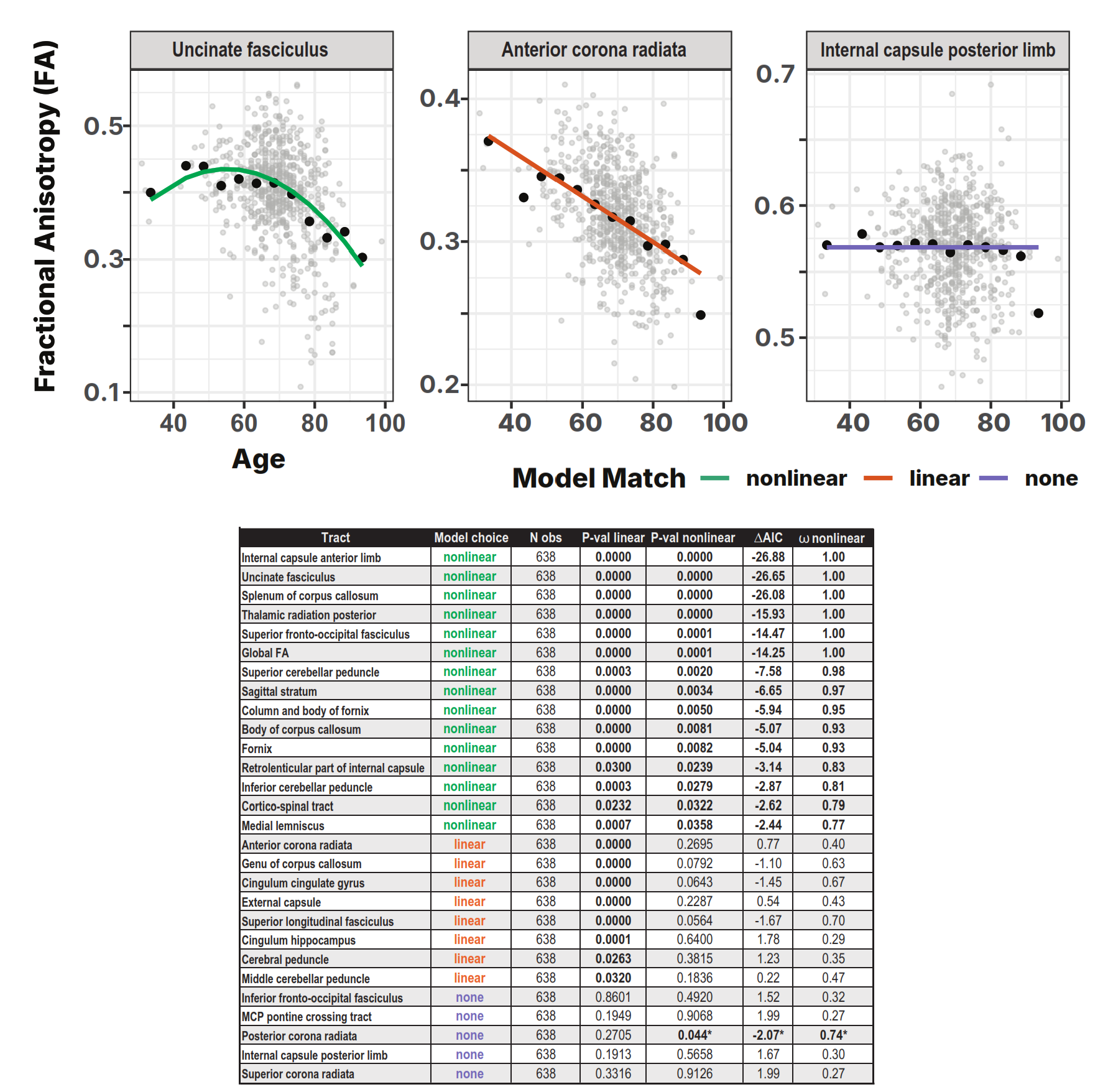
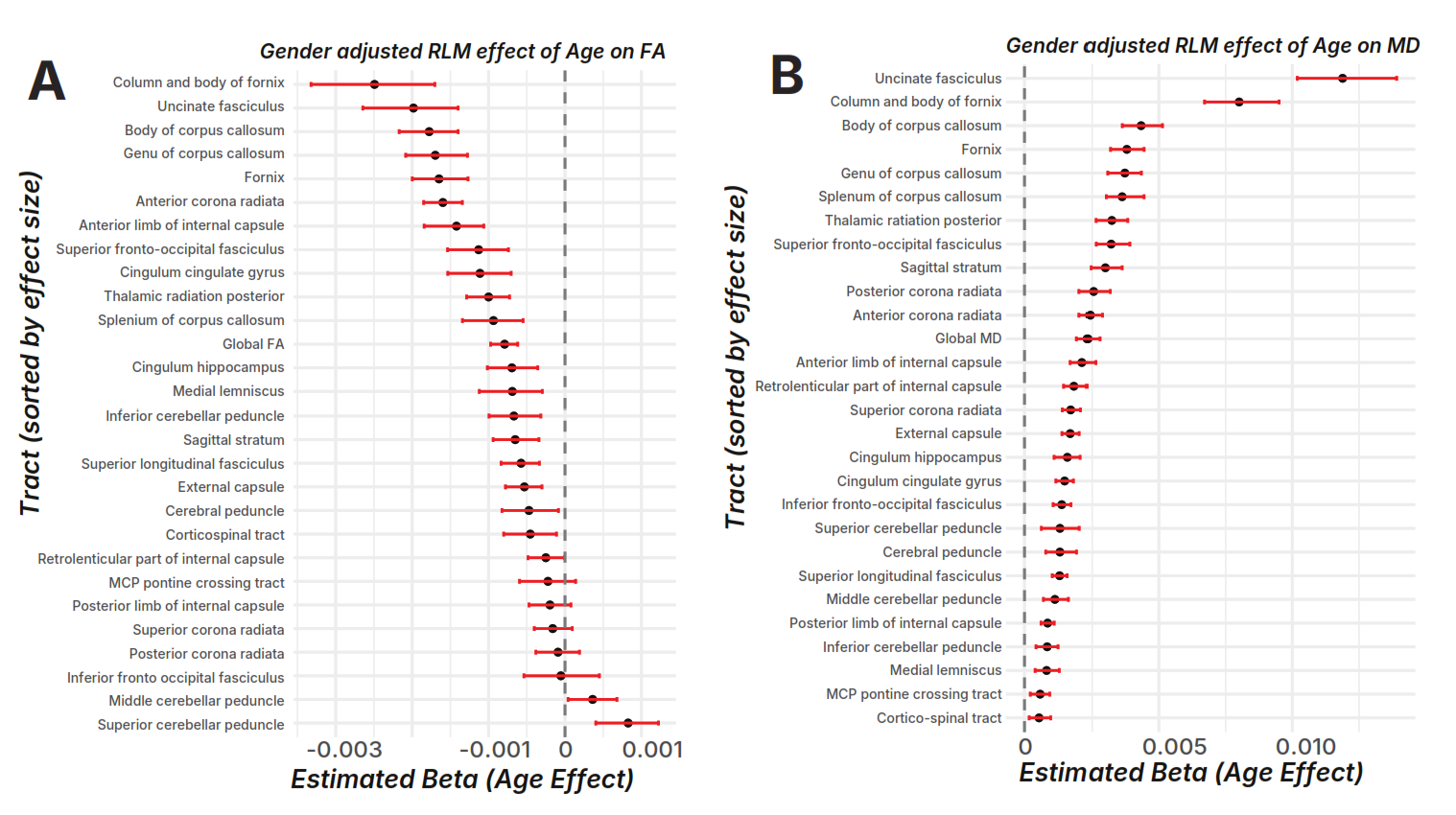
White matter integrity declines heterogeneously with age, with accelerated loss in higher-order association tracts.
Microstructural changes are consistent with myelin loss, not axonal loss.
Microstructure and cognition share a single dominant age-dependent dimension.
Each of the three sections that follow tests one of these predictions and reports whether the data confirm it.
Two complementary metrics from diffusion-weighted MRI were measured across 28 white matter tracts:
Directional coherence of water diffusion. High FA = well-organized fibers. Decreases indicate disorganization and demyelination.
Overall water molecule movement. Low MD = dense, well-myelinated tissue. Increases indicate expanded extracellular space from myelin/cell loss.
Higher-order cognitive tracts (red) show the steepest decline. Sensory/motor pathways (green) are relatively spared. 15 of 28 tracts showed nonlinear (accelerating) decline after ~age 60.
| Tract | Function | FA β | Trajectory |
|---|---|---|---|
| Uncinate fasciculus | Social-emotional, memory, language | −0.0027 | Nonlinear (ω=1.00) |
| Fornix (column & body) | Episodic memory, spatial navigation | −0.0026 | Nonlinear |
| Ant. limb internal capsule | Executive control, decision making | −0.0024 | Nonlinear (ω=1.00) |
| Corpus callosum (body/genu) | Interhemispheric communication | moderate | Mixed |
| Corticospinal tract | Motor control | mild | Linear / minimal |
| Cerebellar peduncles | Motor coordination | mild | Linear / minimal |
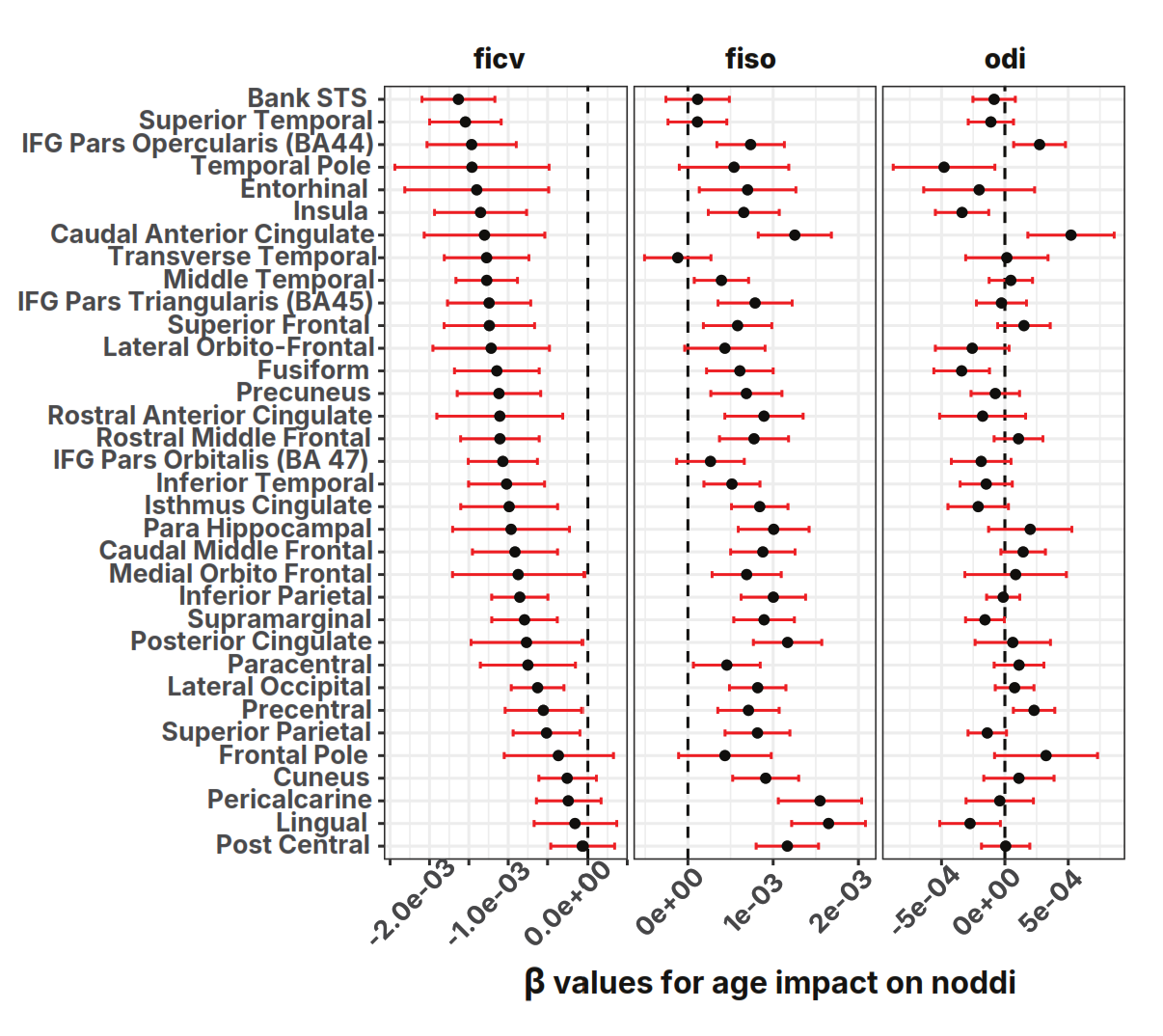
FA/MD conflate multiple tissue properties. NODDINODDIAn advanced MRI analysis separating nerve fiber density from surrounding water.
Full glossary entry → (Neurite Orientation Dispersion and Density Imaging) separates three compartments in white matter "cushions" under 34 cortical regions:
It's the joint signature that matters. A pure axonal-loss process would show ficv↓ together with a change in odi (orientation dispersion) as remaining axons reorganized. The preserved odi rules that out. Demyelination — thinning of the insulation around axons that remain in place — produces exactly this combination: less intracellular signal, more free water, unchanged fiber orientation.
Critical insight: The fiber architecture (odi) is preserved. The brain's wiring diagram remains intact with age. What degrades is the supporting cellular environment: neurite density drops (ficv, likely reflecting oligodendrocyte and myelin sheath loss) and free water fills the vacated space (fiso). In short, the cables are still routed correctly, but their insulation is thinning. This pattern implicates glial/myelin failure rather than axonal death.
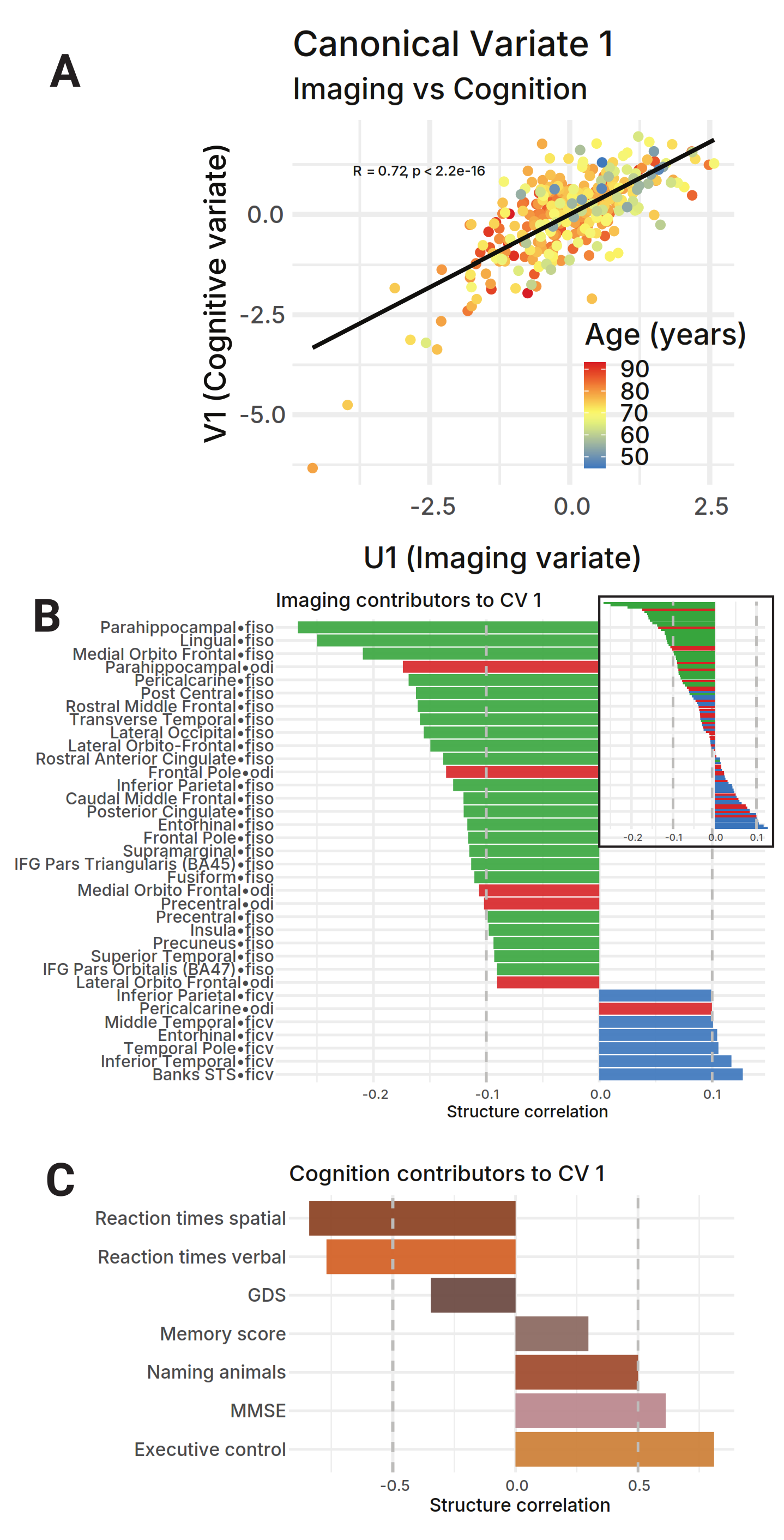
Canonical Correlation AnalysisCCAA statistical method for finding the strongest shared patterns between two datasets.
Full glossary entry → between all NODDI metrics (102 imaging variables) and 7 cognitive assessments revealed a single dominant mode:
Predominantly driven by fiso in association cortex.
Processing speed (measured by reaction times) is the cognitive domain most tightly coupled to white matter microstructural damage. Executive function and global cognition follow. Critically, age is the dominant axis along which structural damage and cognitive decline co-vary: younger participants cluster in the high-integrity / high-performance quadrant; older participants cluster in the opposite corner.
Hillblom Aging Network, UCSF Memory & Aging Center. IRB-approved; written informed consent. 638 complete observations after exclusion, ages 40–99 (N=588 for analyses requiring full NODDI parameterization, after additional QC exclusions).
Siemens Trio 3T or Prisma 3T scanners. MPRAGE T1w: TR/TE/TI = 2300/2.98/900ms (Trio), 2300/2.9/900ms (Prisma); flip angle 9°; FOV 240×256mm; 1mm isotropic; sagittal orientation. Diffusion MRI: multi-shell sampling for DTI and NODDI. Preprocessing: eddy current correction, motion correction, susceptibility distortion correction, B0 field correction. Scanner harmonization: empirical Bayes ComBat across all sessions.
Cognitive–MRI matching: nearest assessment within ±2-year window (temporally closest when multiple available). Sessions missing >35% cognitive variables excluded. Remaining gaps: age-neighborhood median imputation (±5 years, ≥3 observations required). Single visit per patient retained.
Hemispheric averaging: between-subject variability ≫ within-subject laterality; left/right values averaged per tract. Outlier screening: sex-stratified LOESS (span=0.75), IQR residuals, median+3×IQR threshold → entire subject exclusion.
Seven measures: MMSE (global cognition), GDS (geriatric depression), memory z-score (episodic memory), animal naming (semantic fluency), executive/bedside z-score (executive function), verbal reaction time, spatial reaction time (processing speed). Assessments matched within 1-year window of imaging for CCA.
Tensor model: Standard tensor fitting → FA and MD. Harmonized via ComBat. NODDI model: Three retained parameters: intracellular volume fraction (fICV), isotropic free water fraction (fISO), orientation dispersion index (ODI). Parcellation: Atlas-based white matter ROIs; left/right averaged.
Robust Linear RegressionRobust Linear ModelA regression method resistant to outlier data points, using the Huber estimator.
Full glossary entry → (MASS package, R 4.4.1) using the Huber psi-function with iteratively reweighted least squares (IRLS). This M-estimator downweights outlier influence without assuming normality. Because p-values are unreliable under these conditions, statistical inference relies on 1000 nonparametric bootstrapBootstrapEstimating statistical confidence by repeatedly resampling from observed data.
Full glossary entry → iterations yielding 95% percentile confidence intervals. An effect is deemed significant when the CI excludes zero.
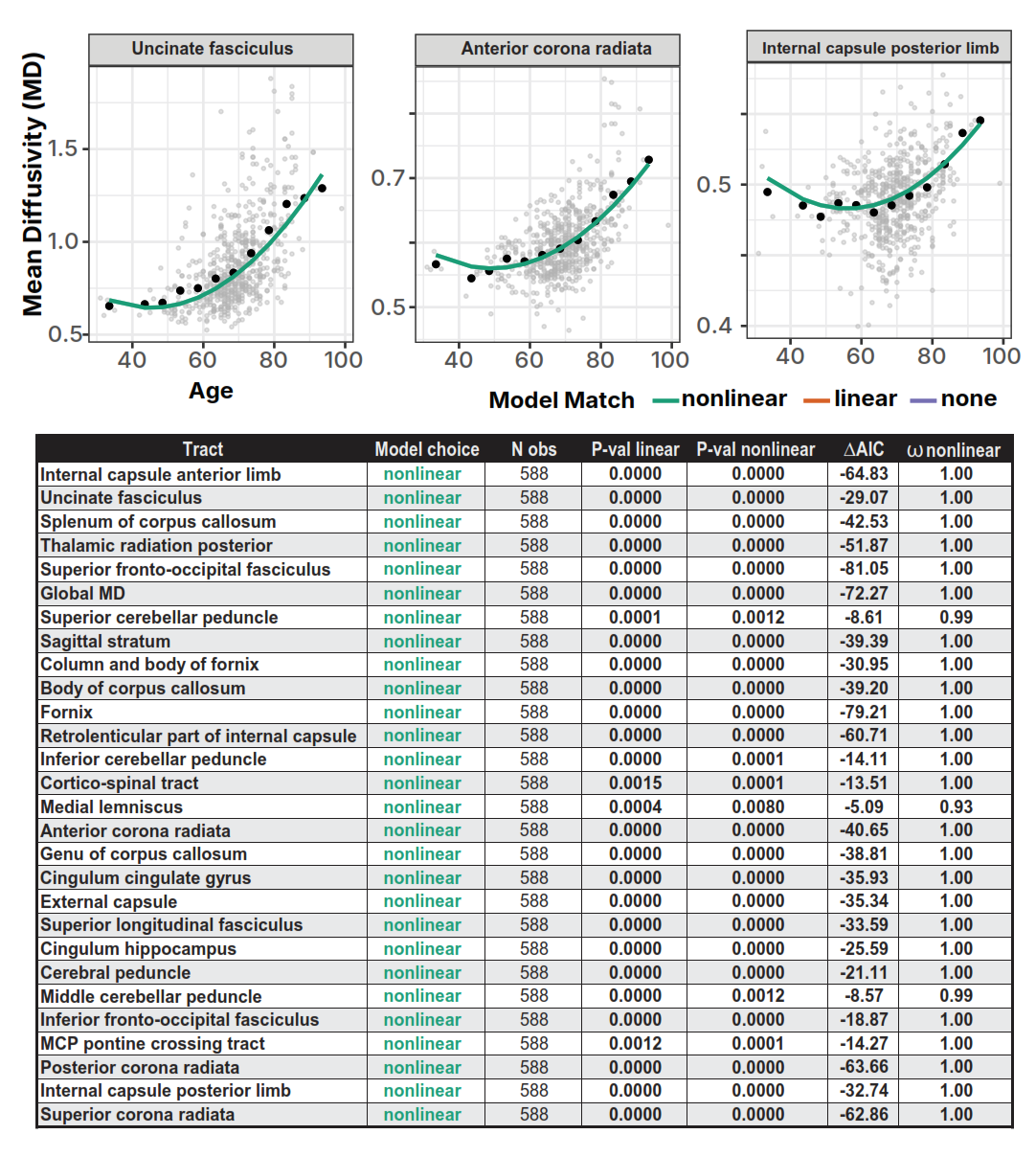
Model selection (linear vs. nonlinear trajectories):
Nested ANOVA F-tests: null vs. linear (Plinear) and linear vs. quadratic (Pnonlinear). Information-theoretic criteria:
Decision rule for retaining quadratic: Pnonlinear < 0.05 AND ΔAIC ≤ −2 AND/OR ωnonlinear ≥ 0.70. Otherwise retain linear if Plinear < 0.05, else classify as "none" (no detectable age effect).
Note: Posterior corona radiata reached nonlinear significance but failed the linear test; visual inspection confirmed outlier-driven → classified as "none."
15/28 tracts showed strong nonlinear evidence (ω > 0.77). 8 tracts best fit linear. 5 tracts showed no significant age effects.
| Tract | βage | ΔAIC | ωNL | Model |
|---|---|---|---|---|
| Internal capsule ant. limb | −0.0024 | −26.88 | 1.00 | Nonlinear |
| Uncinate fasciculus | −0.0027 | −26.65 | 1.00 | Nonlinear |
| Splenium corpus callosum | — | −26.08 | 1.00 | Nonlinear |
| Fornix (column & body) | −0.0026 | — | >0.77 | Nonlinear |
| Anterior corona radiata | — | — | — | Linear |
| Genu corpus callosum | — | — | — | Linear |
| Internal capsule post. limb | — | — | — | None |
MD trajectories: All 28 tracts showed significant nonlinear relationships (ω ≥ 0.93). Universal J/U-shaped pattern confirmed by 5-year bin averaging. Strongest: internal capsule ant. limb (ΔAIC = −64.83), splenium (ΔAIC = −42.53), uncinate fasciculus (ΔAIC = −29.07).
Robust linear models across 34 cortical white matter cushion parcels:
Interpretation: Fiber architecture preserved; cellular environment degrades. Consistent with oligodendrocyte/myelin loss > axonal degeneration. Spatial heterogeneity in ficv/fiso sensitivity recapitulates tract-level FA/MD hierarchy.
Design: X = 34 parcels × 3 NODDI metrics (102 variables). Y = 7 cognitive measures. Both standardized (zero mean, unit variance). Canonical variates computed in standardized form. Structure correlations = cor(X, U) and cor(Y, V). Significance: Wilks' lambdaWilks' LambdaA test statistic for determining if CCA results are statistically significant.
Full glossary entry → F-test (yacca). Age regression on all variates with FDR correctionFDR CorrectionA method to reduce false positives when running many statistical tests.
Full glossary entry →.
Age loading on CV1: U1 ~ Age: β = 0.041 ± 0.002 (p < 0.001). V1 ~ Age: β = −0.058 ± 0.003 (p < 0.001). Age is the dominant axis of the brain–cognition relationship in this cohort.
Supplementary CCA Analysis (Fig. S3): Scree plot confirms CV1 as the only significant dimension (R²=0.52). Age regression with FDR correction shows U1 (β=0.041, p<0.001) and V1 (β=−0.058, p<0.001) are both age-dependent. Cross-domain correlation matrix reveals fiso measures have the highest correlations with cognitive variables. Within-imaging correlations show little structure within each NODDI model, confirming that CCA captures genuinely multivariate relationships. Full supplementary panels available in the preprint.
Three developmental waves of myelin change (establishment in childhood, maturation in adolescence, atrophy in aging) map onto three major classes of neurological disease: neurodevelopmental, psychiatric, and neurodegenerative, respectively. Genes associated with all three disease classes are enriched in oligodendrocyte lineage cells, suggesting that myelin biology is a shared vulnerability axis across the lifespan.
The activity-dependent feedback loop that maintains myelin:
Age-related breakdown of this loop (e.g., via inflammatory signaling to OPC/OL populations) cascades into progressive decoherence of functional networks. This positions myelin maintenance as a potential intervention point.
Six numbers that frame what the brain's hidden infrastructure does, and what happens when it fails.
This theory opens doors for different communities. Find yours.
The full preprint with methods, data tables, and supplementary figures will be available on bioRxiv. We welcome replication, extension, and collaboration.
@article{gershteyn2026ddh,
title = {Decoherence via Demyelination (DDH): A Hypothesized Mechanism of Cognitive Decline},
author = {Gershteyn, Iosif M. and Markov, Nikola T. and Kramer, Joel and
Casaletto, Kaitlin and Ellerby, Lisa M. and Furman, David},
journal = {bioRxiv},
year = {2026},
note = {Preprint. DOI to be assigned upon posting.},
url = {https://ddh-theory.com}
}
DOI will be added once the preprint posts to bioRxiv. Please check back or subscribe below for the launch notification.
Corresponding authors iosifmg@gmail.com nmarkov@buckinstitute.org dfurman@buckinstitute.org
The DDH identifies myelin maintenance as the primary structural target for cognitive aging — complementary to amyloid- and tau-focused programs, and capable of integrating gains from neurovascular, anti-inflammatory, and lifestyle interventions. Key areas of opportunity:
If you or someone you love is experiencing age-related cognitive changes, here's what this research suggests:
This is an active area of research. Consult your physician for personalized medical advice.
The DDH reframes cognitive aging and opens new therapeutic directions.
Prevailing models of cognitive aging emphasize neuronal death and synaptic degradation. The DDH proposes a different primary driver: loss of inter-regional timing precision caused by heterogeneous myelin degradation across white matter pathways.
This reframes the therapeutic target. Rather than trying to keep individual neurons alive, interventions can aim to preserve or restore the connective infrastructure: the myelin that synchronizes distributed brain networks.
One question the DDH has to answer: if white matter changes are this dramatic, why don't most people experience catastrophic cognitive decline as they age? For the majority, the deficits are mild.
The likely explanation is compensation. The aging brain recruits additional functional connectivity, over-activates certain circuits, and re-routes around damaged pathways to preserve performance on familiar tasks. Studies of normally developing children compared with children who have demyelinating disorders point to changes in inter-areal synchronization as a core compensatory mechanism.
The DDH suggests a sobering corollary: because demyelination is heterogeneous, it produces asymmetric damage across functional sub-networks. This asymmetry can outpace the brain's ability to "re-equilibrate" through compensation, helping explain why some pathways (and the cognitive functions they serve) decline before others, and why decline accelerates after age 60.
Practical implication: cognitive engagement, novel learning, exercise, and sleep all support the activity-dependent feedback loops that maintain myelin and recruit compensation. None is a cure. All are more than nothing.